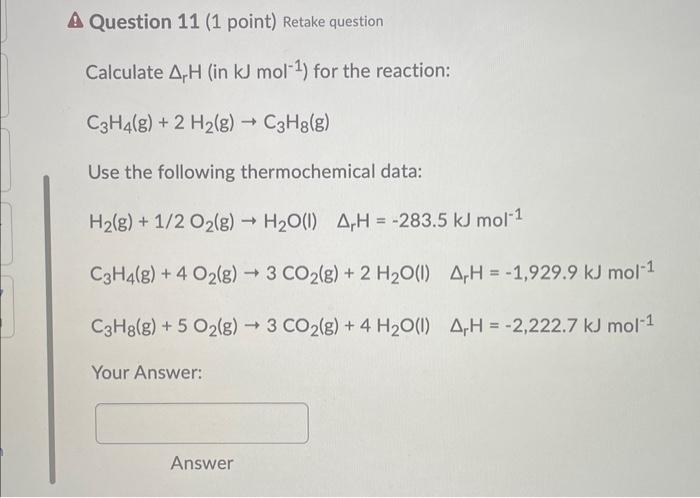
SOLVED: Given the data: N2(g), ΔH° = 0.00 kJ mol-1, ΔS° = +191.5 J mol-1 K-1 Hz(g), ΔH° = 0.00 kJ mol-1, ΔS° = +130.6 J mol-1 K-1 NH3(g), ΔH° = -46.0

∆H 0 m (kJ mol −1 ) and ∆S 0 m (kJ mol −1 K −1 ) for the SDS-caffeic... | Download Scientific Diagram

enthalpy - Why is it sometimes kJ only, and in other times kJ/mol? What's the difference? - Chemistry Stack Exchange
13. Enthalpy and entropy changes of a reversible reaction are 40.63 kJ mol–1 and 108.8 J K–1 mol–1 respectively. Predict the feasibility of the reaction at 27^° C.

The standard enthalpy of formation of NH, is -46.0kJ mol"!. If the enthalpy of formation of H, from its atoms is -436kJ mol-' and that of N, is –712kJ mol-', the average

66. CH + 50, +300, + 4H,0 AH =-2220 kJ moF1 CH+H, CH, AH =-124 kJ mol-1 H+ +0, H,0 AH = -285 kJ mol-1 Then heat of combustion of CH (1)-2059 kJ mol-1 (2) 2059 kJ mo (3) -4118 kJ mol- (4) +4118 kJ mol-1
The activation energy of a reaction is 75.2 kJ mol^-1 in the absence of a catalyst and it lowers to 50.14 kJmol^-1 with a catalyst. - Sarthaks eConnect | Largest Online Education Community

enthalpy - Why is it sometimes kJ only, and in other times kJ/mol? What's the difference? - Chemistry Stack Exchange

Enthalpy of formation of H,O, is: (1) +X, kJ mol-1 127-X, kJ mol (3) +X, kJ molt (4) -X, kJ mol Given that bond energies of H-Hand CHCl are 430 kJ moll
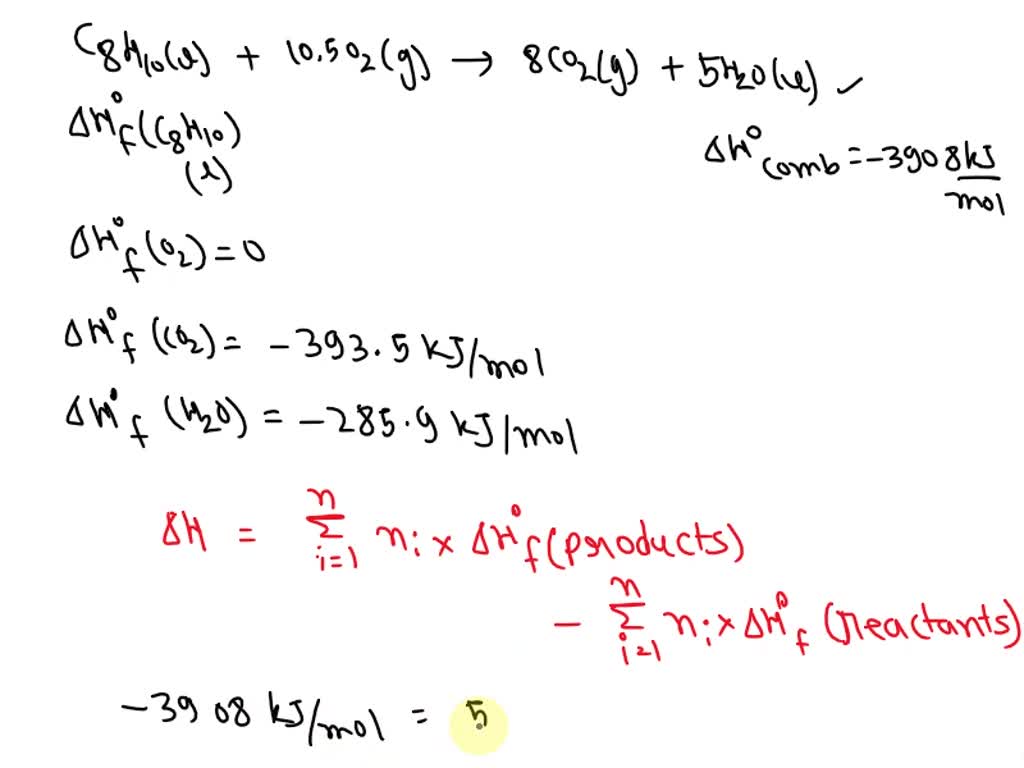
SOLVED: Physical chemistry Knowing that the standard enthalpy of combustion for xylene, CgH10(I), is -3908 kJ mol-1. Using HOf: H2O(l) = -285.9 kJ mol-1; CO2(g) = -393.5 kJ mol-1. CgH10(l) + 10.5O2(g)
According to the following figure, the magnitude of the enthalpy change of the reaction A + B → M + N in kJ mol^–1 - Sarthaks eConnect | Largest Online Education Community

Calculated BDEs and BDEs in (kJ mol -1 ) of ortho-and meta-substituted... | Download Scientific Diagram